
1. H2 and its impact on UFCC performance
Among the products generated in the catalytic cracking unit, hydrogen (H2) is part of the components of the fuel gas stream, along with methane, ethane, and ethylene.
Although it is a product that is part of the UFCC outlet streams, the increased concentration of H2 in the fuel gas can be a problem, as it decreases the molecular weight of the wet gas, generating changes in the compression ratio and displacement of the operating point of the performance curve, directly affecting the performance of the wet gas compressor.
This represents a challenge for the technical team, which must ensure the operation of the wet gas compressor, managing integrity risks for the equipment while also making adjustments to reduce the impacts this has on the products with higher added value.
2. Heavy metals and their contribution to dehydrogenation
An increase in H2 generation is usually directly related to the action of nickel in the system. Although other metals also have dehydrogenating action, their effects are less significant compared to those of nickel.
Nickel (Ni) is found in the feedstock, in the form of molecules called porphyrins which are deposited on the catalyst, and in the converter environment it’s reduced to its metallic form Ni0, acquiring greater mobility, catalyzing the dehydrogenation reactions.
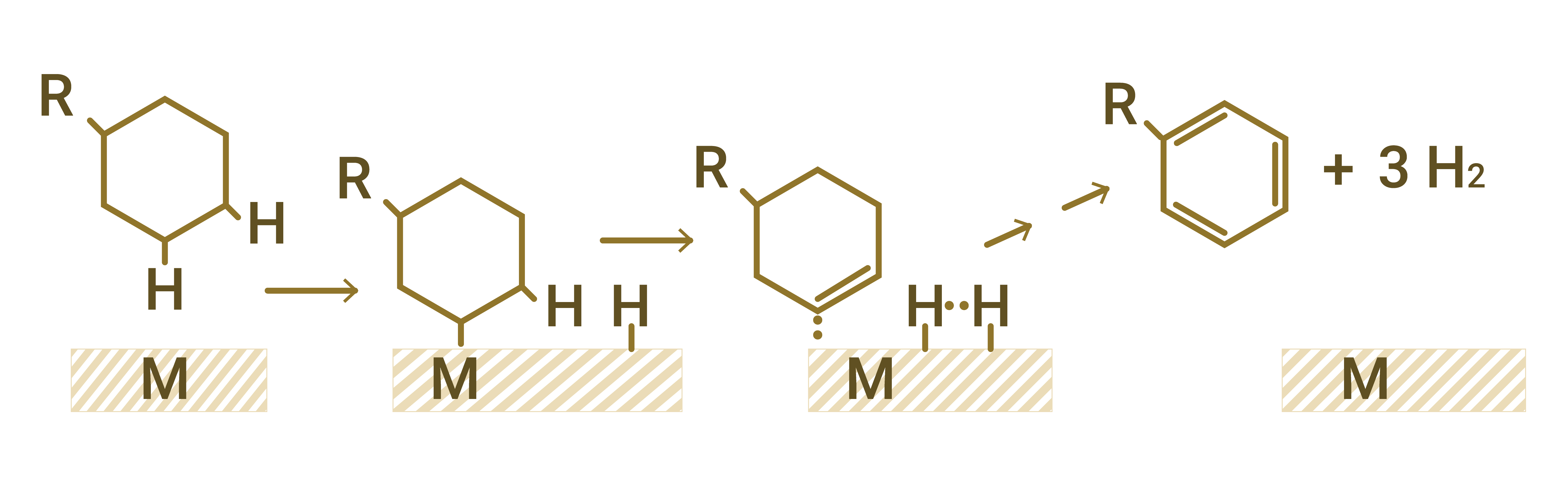
Figure 1 - Dehydrogenaating effect of transition metals, such as nickel. Source: Database of FCC S.A.
Dehydrogenation reactions, in addition to leading to an increase in H2 concentration in the products, cause the condensation of hydrocarbons that ultimately form coke. That is why it is common for the refineries facing issues with excess H2 in the gas to also be experiencing a period of increased coke yield.
Once these reactions begin, an increase in the H2/CH4, ratio will be observed, indicating a change in the quality of the wet gas due to the ongoing dehydrogenation action. This property, normally monitored by process engineers, should require a rigorous control and monitoring strategy when a gradual increasing trend is observed starting from 0.3%v/%v and the need for mitigating actions when 0.5%v/%v is reached.
According to Letzsch, 2024, a normal production of H2 per barrel of feedstock processed should range between 20 and 45 scfb (3.56 and 7.99 sm3/m3), starting from 60 scfb (10.69 sm3/m3) passivation mechanisms are viable, becoming mandatory when values reach 100 scfb (17.83 sm3/m3). However, these reference values may vary slightly depending on the design of each unit.
The graph below shows the increasing trend of nickel in feedstock accompanied by an increase in the H2/CH4 ratio, reaching values that strongly indicate an increase in dehydrogenation reactions.
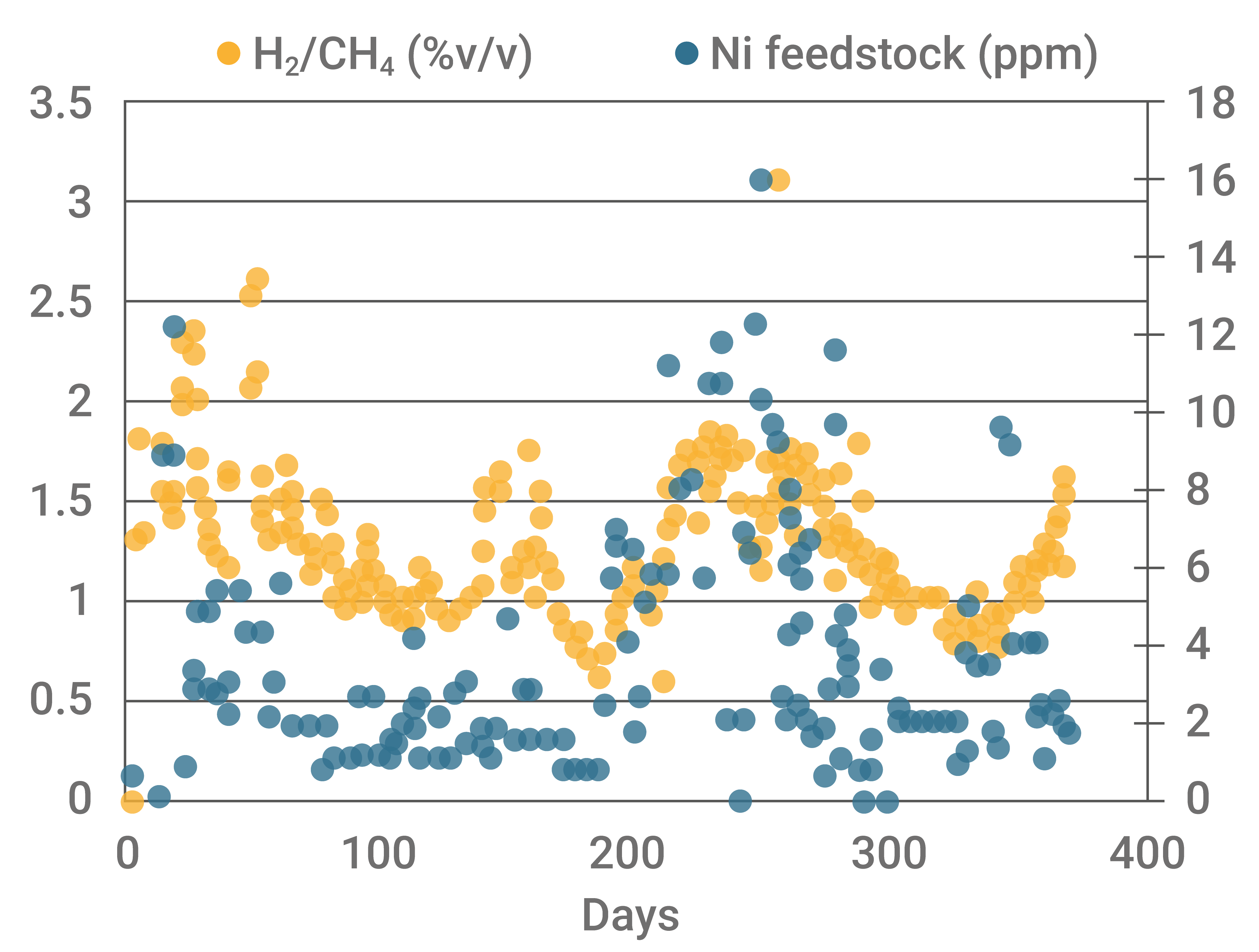
Graph 1 - Response of the H2/CH4 ratio to changes in nickel concentration in the feedstock. Source: Database of FCC S.A.
After nickel, vanadium is another metal that contributes to dehydrogenation due to its ability to interact with it.
The dehydrogenating action of vanadium compared to nickel is 25%. The mechanism is due to vanadic acid (H3VO4) reacting with nickel, forming nickel vanadate (Ni2V2O7) as the final product, which deposits on the catalyst and promotes dehydrogenation (Etim et al., 2016). The combined effect of both Ni and V, is referred to as equivalent nickel. This parameter is calculated based on the content of contaminants in the Ecat, allowing monitoring and cross-referencing of data on the combined effect and increase of H2, allowing us to follow its activity in the system.
The following graph shows the correlation of the trend between the increase of equivalent nickel with the H2/CH4 ratio of the unit. The higher dehydrogenating action at Nieq peaks in the inventory is clear.
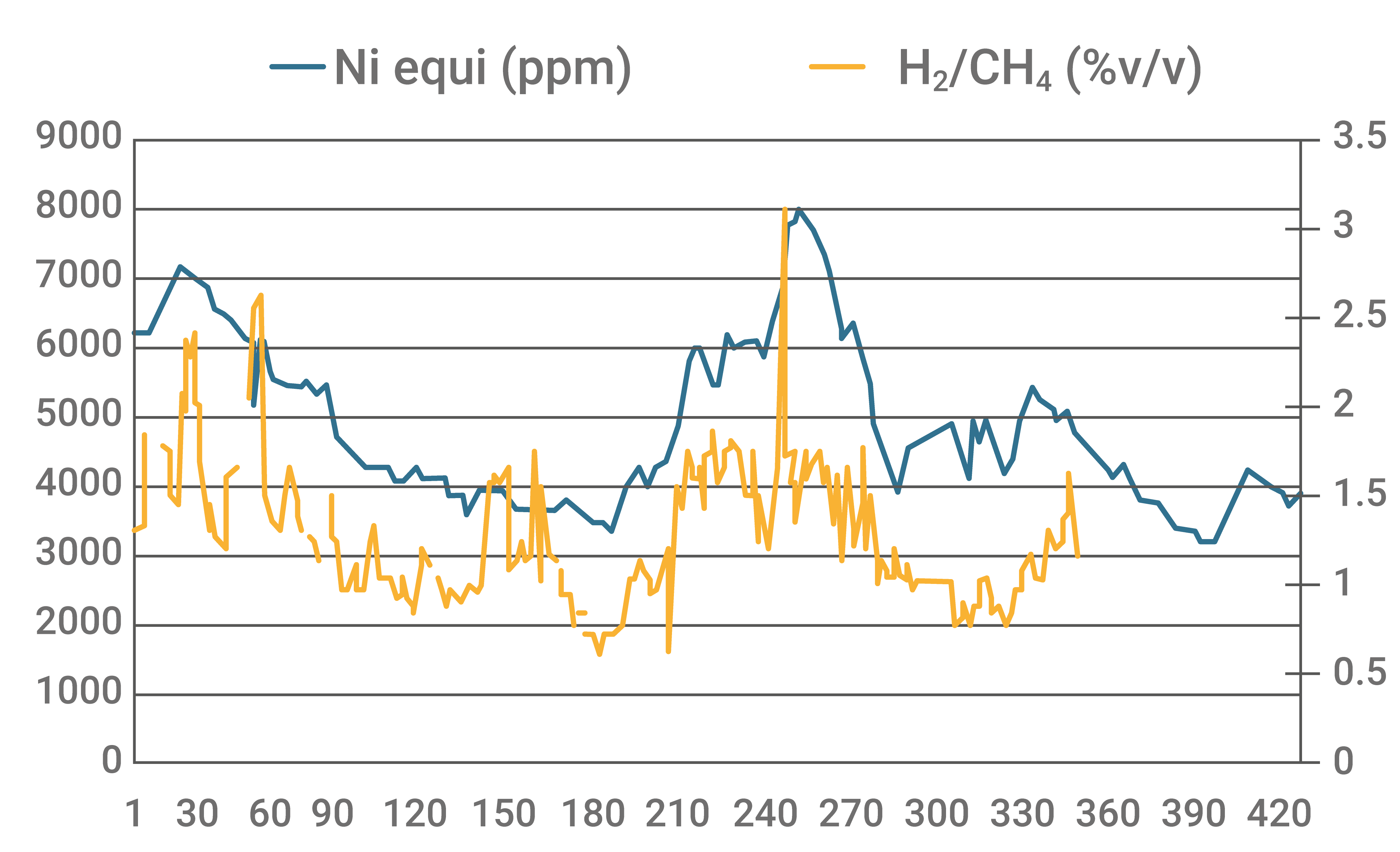
Graph 2 - Variation of the H2/CH4 ratio with changes in the equivalent nickel concentration in the inventory. Source: Database of FCC S.A.
The dehydrogenating effect of other metals can be summarized as follows: the dehydrogenating action of iron is 10 times lower when compared to nickel. Copper’s is 3 times lower. Both also have an impact on coke production.
3. Consequences of the increase in dehydrogenation reactions
In an FCC unit, the liquid products with the highest commercial value have a hydrogen-to-carbon (H/C) ratio greater than that of the feedstock. The acidic characteristic of the catalyst not only promotes chain-breaking reactions of hydrocarbons but also promotes the stabilization of catalytic cracking products through proton donation and hydrogen transfer reactions, resulting in hydrocarbons with lower molecular weight. Once there is a limited supply of hydrogen atoms in the system, secondary reactions occur and form hydrocarbons with higher molecular weight and a low H/C ratio, which is coke. That is the hydrogen balance.
The mechanisms that promote the formation of H2 result in less hydrogen being available to stabilize hydrocarbons. The consequence is the formation of more unstable compounds, such as olefins, and more hydrocarbons with low hydrogen content, such as coke. That excess coke, not related to catalytic cracking reactions, interferes with the thermal balance of the unit. This is because the coke mass produced in the riser/reactor system must be the same as that burned in the regenerator. When there is a mechanism that increases coke production unrelated to catalytic cracking, there must be compensation for the reduction in catalytic coke production, that is, a reduction in conversion occurs.
The increase in the concentration of H2 in the fuel gas can also lead to operational difficulties in the gas recovery and treatment section, in addition to potentially affecting equipment outside the FCC unit.
The most notable consequence of the refinery is the change in the operating point of the wet gas compressor. The reduction of the molecular weight of the fuel gas and the consequent increase in specific volume cause the compressor’s rotation to increase and its efficiency to change, leading many systems to operate closer to the surge curve. That is usually the ocasion when investigations concerning the fuel gas begin.
Another possible consequence of the increase in specific volume of the fuel gas is making its treatment harder. Absorption columns with sponge hydrocarbons and amine treatment columns can experience flooding due to excessive volumetric gas flow, leading to liquid carryover and the presence of excess contaminants such as H2S and LPG fractions.
Finally, the change in composition affects the heating value of the fuel gas, which decreases in volumetric terms and causes an increase in the opening of the gas flow control valves of the burners. In addition to potential flow limitation problems for some of these burners, this can affect the energy balance calculations of some refineries because not all of them update the calculations of that balance from fuel gas chromatography results; they do so by using an average composition and a standard heating value.
4. Dehydrogenation control mechanisms - Passivation
One of the ways to reduce the concentration of H2 in the fuel gas is by controlling the dehydrogenating action of nickel through its passivation, which can be achieved through two main strategies:
- Use of catalysts formulated with nickel trap;
- Use of antimony pentoxide (Sb2O5) to passivate nickel;
The function of a trap is to reduce the dehydrogenating action of nickel, by oxidizing it to Ni+2. With that reduction, there will consequently be a decrease in the delta coke. It is key that the addition rate is calculated based on the concentration of the contaminant in the feedstock to ensure that there will be less active nickel available, thus reducing its mobility and action.
Fábrica Carioca de Catalisadores S.A.’s catalyst technologies include MT-60 in its formulations, a high-performance nickel trap.
Antimony pentoxide is an additive used globally for the passivation of nickel, forming a stable alloy with the metallic form Ni0, creating a barrier so that nickel cannot react withhydrocarbons. The rate of injection of this additive is based on the concentration of nickel in the feedstock.
It is important to consider that a high injection of antimony pentoxide can lead to increased NOx emissions and may promote deposition and fouling in the bottom circuit of the main fractionator. Hence the importance of ensuring that the Sb/Ni ratio remains within the range of 0.20–0.40, with the possibility of working with lower ratios when the catalyst has nickel traps. Excess antimony pentoxide can also partially passivate combustion-promoting additives based on platinum.
5. Control mechanisms to reduce H2 concentration in fuel gas - Operational
Operationally, there are some adjustments that the technical team can make in order to reduce the concentration of H2 in the fuel gas. Some of the main ones are:
- Control the nickel concentration in the feedstock: establishing a maximum concentration helps to avoid operational upsets;
- Control the chloride concentration in the feedstock due to it’s capacityto reduce Ni+2 to Ni0: the reactivation of nickel in the catalyst can lead to a significant increase in dehydrogenation reactions;
- Increasing the reaction temperature: it does not seem like a logical change, since the fuel gas yield increases, but by favoring catalytic cracking reactions, the increase in reaction temperature reduces the concentration of H2 in the fuel gas, as shown by the example obtained through a process simulator:
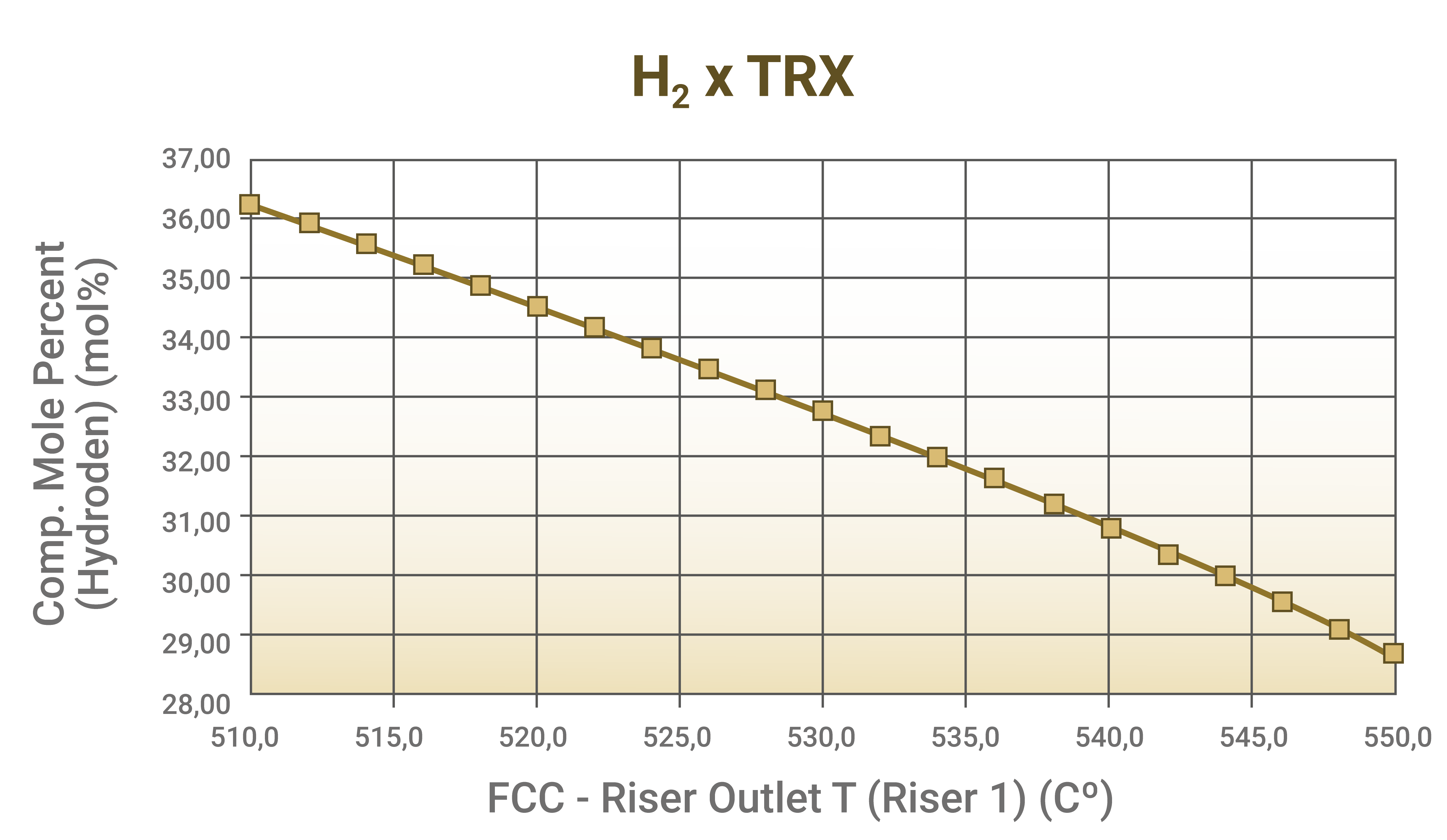
Graph 3 - Reduction of H2 concentration in the fuel gas through increased reaction temperature. Source: Results of simulations conducted by FCC S.A.
- Increasing feed temperature and improving feedstock dispersion: inadequate vaporization of the feedstock causes the higher molecular weight hydrocarbons to remain liquid when they come in contact with the catalyst, leading to coke formation and reduced availability of active sites at the lower portion of the riser. That phenomenon increases the available time for condensation and dehydrogenation reactions to occur.
The increase in feedstock temperature, despite reducing catalyst circulation and potentially increasing fuel gas yield, can be used to reduce the concentration of H2 in the fuel gas, especially in units that operate with residual feedstock, which may not fully vaporize at low temperatures.
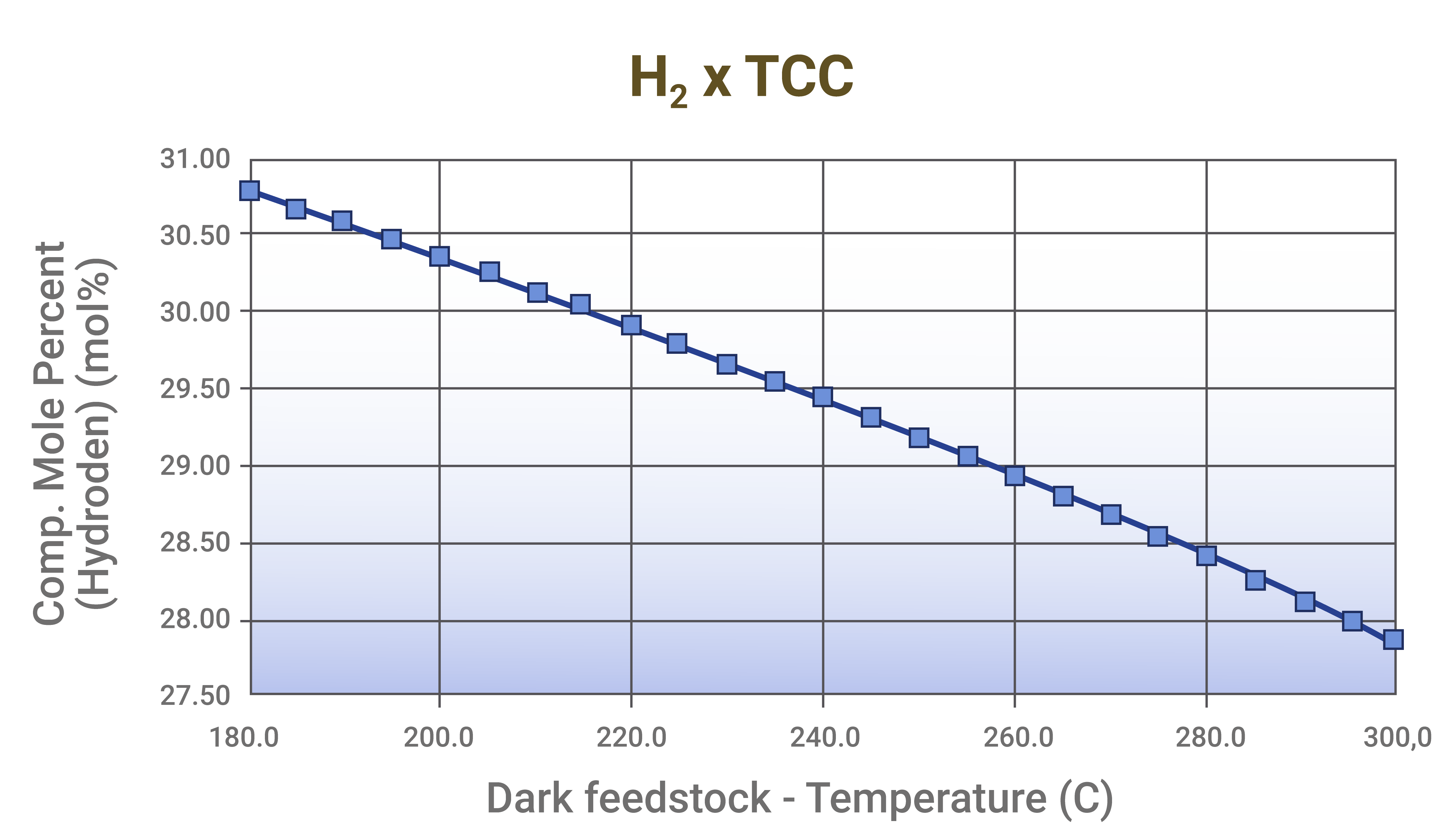
Graph 4 - Reduction of the H2 concentration in the fuel gas as a function of the increase in the feedstock preheating temperature. Source: Results of simulations conducted by FCC S.A.
It is important to ensure, in addition to adequate feed temperature according to the quality of the feedstock, that the atomization steam flow is aligned with the design requirements of the nozzles and the feedstock flow. This contributes to reducing the size of the oil droplet (the smaller, the better), which will decrease the contact time for vaporization to occur along the riser, favoring the catalytic cracking reactions.
- Increasing fresh catalyst makeup: especially in cases where the fresh catalyst has a nickel trap, increased fresh catalyst makeup can benefit the unit through increased nickel removal from the system (catalyst discard) and greater availability of sites that can react with nickel for passivation.
6. Catalytic solutions
Fábrica Carioca de Catalisadores S.A. offers integrated solutions that can help in challenging times, especially when it comes to scenarios where more contaminated feedstock is part of the projections. The UPGRADER, Denali and Safeguard technologies were developed for heavy and/or contaminated feedstock processing scenarios, delivering unmatched results in conversion and resistance to metals. Each client has a specific catalyst formulation to meet their needs, and the customization of the nickel trap is one of the possible adjustments in those formulations.
Conclusion
Continuous monitoring of the quality of the feedstock and the composition of the fuel gas is key to avoiding operational difficulties caused by excessive increases in H2 concentration in that product. The combined use of feedstock quality control strategies, adequate catalyst formulation, catalyst makeup, operational control, and the eventual use of additives allows refineries to keep the generation of H2 in the UFCC under control.
The technical team at Fábrica Carioca de Catalisadores S.A. has the experience and availability to support our clients in these challenges. The analysis of operational disturbances, the optimization of the unit, and the continuous updating of the catalytic system are part of our Core Services; our services go beyond supplying catalyst.



































What did you make of the publication?